A large collaboration led by researchers from the University of Wollongong has used the Australian Synchrotron to investigate a promising carbon coated cathode material for rechargeable sodium ion batteries that had a very long life.
The research demonstrated that a new compound, an iron-based pyrophosphate-based sodium composite, could compete with other iron-based cathode materials for large scale applications, such as wind and solar storage, because its charge retention capacity was close to 90 per cent after 1100 cycles at 5 C.
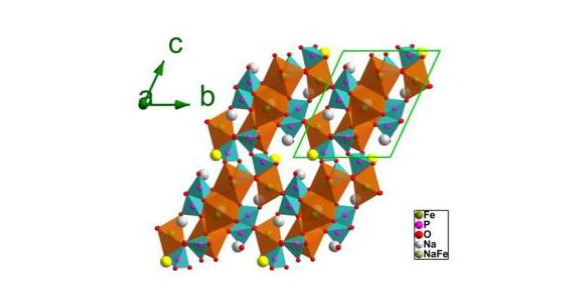
Low energy density, sluggish sodium kinetics and poor cycling stability have been the main barriers to the widespread use of sodium ion batteries as less costly alternative to lithium batteries.
The synchrotron was used to investigate the electrochemical mechanism at work in a new cathode material, Na3.32Fe 2.34 (P2O7) as reported in Advanced Materials.
The multiple anions of the new compounds give them high structural and thermal stability with only small volume changes in the unit cell.
Qinfen Gu, a senior scientist on the powder diffraction beamline, said it was possible to charge and discharge the battery in real time while sending X-rays through the nanoparticles, which revealed what was happening at a structural level in the cathode material during cycling.
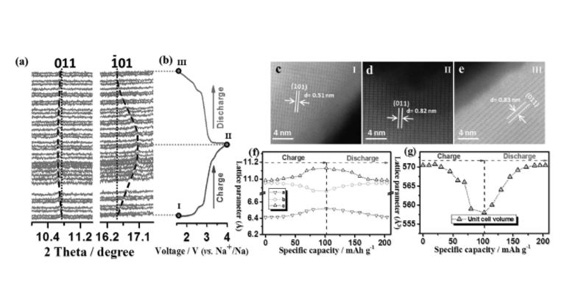
The diffraction data indicated a single phase transition during charge and discharge, which helped explain its superior electrical performance.
A reversible electrochemical reaction, suggested by continuous lattice breathing, also took place in the material.
“The movement of the sodium ions can cause the cathode to expand or contract, which can degrade the material and interfere with the movement of electrons,” said Gu.
In this case the researchers noted that only a small change in lattice parameters and unit cell offered strong support for the stability of the structure during cycling.
The application of a carbon coating on the cathode material improved its thermal and structural stability, enhanced its electrical conductivity and contributed to a high rate capacity.
The investigators also attributed superior electrochemical performance to a dramatic increase in the transfer rate of electrons across different layers of the material and an acceleration of the transfer of sodium ions by the uniform amorphous carbon layer.
Experiments confirmed that without the carbon coating, the material degrades very quickly and after 500 cycles, charge retention capacity drops.
“The electrolyte attacks the cathode material and the carbon coating slows this down,” explained Gu.
The material would be low cost because it is primarily composed of ample elements, sodium and iron, and offers improved safety.
Other collaborating institutions included the University of South China, the University of Queensland and Sichuan University (China).
