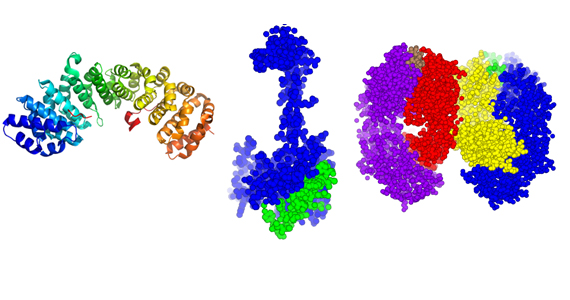
Australian research into the molecular make-up of cells in the body, common elements, and minerals has reached a milestone with researchers at the Australian Synchrotron releasing detail of their 1000th protein structure to the world, paving the way for improved understanding of disease and more targeted therapies.
Detailed imagery of the Bax protein (pictured, centre), determined by scientists from the Walter and Eliza Hall Institute of Medical Research in Melbourne using the Australian Synchrotron, was the 1000th structure submitted to the Protein Data Bank, a free and open-access central repository for crucial molecular information that supports global biological research.
Dr Peter Czabotar from the Walter and Eliza Hall Institute says Bax will now be analysed to understand key steps involved in programmed cell death in diseases.
‘Through our experiments at the Australian Synchrotron, we can now picture the molecular structure of Bax and, by analysing its surface, shape and interactions, we can now work toward treatments that support, or block, its activity, depending on its role in different diseases, including cancer.’
Scientists from Australian and New Zealand used the Australian Synchrotron to solve 1,000 protein structures in less than eight years, using only two of the facility’s ten experiment stations, known as beamlines, a rate on par with other, larger synchrotrons in North America, Europe and Asia.
Dr Tom Caradoc-Davies, Principal Scientist of the Macro and Micro-molecular Crystallography (MX1 and MX2) beamlines at the Synchrotron, says the facility is crucial to understanding the structure of proteins, at a molecular level, which cannot be visualised in any other laboratory setting.
‘To reveal a protein’s structure, which may hold the key to better understanding its role in diseases, treatments or industrial products, researchers must purify the protein and turn it into a crystal -- the right crystal can take months to make and may be too small or weak to subject to regular laboratory X-ray research.
‘The Australian Synchrotron’s X-ray light is a million times brighter than the sun, enabling light to diffract off crystals smaller than one-tenth the thickness of a human hair, leading to high definition data sets that can produced in a matter of minutes, rather than days.’
Dr Caradoc-Davies says structures discovered using the Australian Synchrotron have unlocked innovation across a range of scientific fields including medical research, electronics and mining.
‘New appreciation of how proteins are shaped enables scientists to understand their role in the onset and progression of diseases, design novel drugs that target proteins for new medicines, or rationally engineer new medical products.’
Also pictured: the 500th protein deposited to the Protein Data Bank, called 3ZIN (left), solved in 2013 by researchers at The University of Queensland and, (right), a sphere model of Solanezumab, showing every individual atom; insight into how Solanezumab interacts with brain proteins associated with the development of Alzheimer’s highlights what makes current therapies for the disease effective, and show how these therapies can be improved, revealed earlier this year by research from St Vincent’s Institute of Medical Research.